Lab 9 - Equilibrium and Le Châtelier's Principle
Purpose
To observe systems at equilibrium, and to determine what happens when stresses are applied to such systems.Goals
-
•To observe the effect on equilibrium of adding or removing products and reactants.
-
•To predict the direction in which the equilibrium shifts upon a change in concentration of one of the components.
-
•To determine the thermicity of a reaction based on equilibrium shifts.
Introduction
Many chemical systems are considered to be reversible. For example, drop the temperature of water to 0°C and it freezes; raise the temperature above 0°C and it melts. Many chemical reactions are also reversible. If one mixes ammonia and oxygen, the products form according to equation 1:( 1 )
4 NH3(g) + 3 O2(g) → 2 N2(g) + 6 H2O(g)
( 2 )
2 N2(g) + 6 H2O(g) → 4 NH3(g) + 3 O2(g)
( 3 )
4 NH3(g) + 3 O2(g) 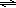 2 N2(g) + 6 H2O(g)
2 N2(g) + 6 H2O(g)
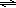 2 N2(g) + 6 H2O(g)
2 N2(g) + 6 H2O(g)-
Products are the chemical species to the right of the equilibrium arrow, as the reaction equation is written.
-
Reactants are the chemical species to the left of the equilibrium arrow, as the reaction equation is written.
-
The forward reaction is the process as written from left to right in the reaction equation.
-
The reverse reaction is the process as written from right to left in the reaction equation.
4 NH3(g) + 3 O2(g) 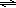 2 N2(g) + 6 H2O(g)
2 N2(g) + 6 H2O(g)
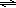 2 N2(g) + 6 H2O(g)
2 N2(g) + 6 H2O(g)-
If a stress is applied to a system at equilibrium, the system will respond by shifting in the direction that reduces the stress.
4 NH3(g) + 3 O2(g) 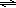 2 N2(g) + 6 H2O(g)
2 N2(g) + 6 H2O(g)
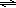 2 N2(g) + 6 H2O(g)
2 N2(g) + 6 H2O(g)4 NH3(g) + 3 O2(g) 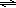 2 N2(g) + 6 H2O(g)
2 N2(g) + 6 H2O(g)
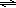 2 N2(g) + 6 H2O(g)
2 N2(g) + 6 H2O(g)( 4 )
| Fe3+ + SCN− | 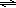 | FeSCN2+ |
| colorless | 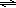 | red |
( 5 )
| CoCl42− + 6 H2O | 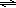 | Co(H2O)62+ + 4 Cl− |
| blue | 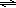 | pink |
Equipment
-
2 30 mL beakers
-
1 100 mL beaker
-
1 ceramic spot plate
-
2 glass stir rods
-
1 hot plate
-
1 250 mL beaker for waste collection
-
1 deionized water squirt bottle
Reagents
-
~3 mL 0.010 M Fe(NO3)3
-
~1 mL 0.10 M Fe(NO3)3
-
~1 mL 0.05 M NaSCN
-
~0.1 mL 1.0 M AgNO3
-
~0.1 mL 1.0 M NaNO3
-
~15 mL 0.10 M Co(NO3)2 (plus some in a dropper bottle)
-
? mL 12 M HCl
-
ice
Safety
Concentrated hydrochloric acid (12 M HCl) is very corrosive, and its vapor is a respiratory irritant. Work with it under the fume hood and avoid inhaling the vapor. Liquid hydrochloric acid can attack the skin and cause permanent damage to the eyes. If it splashes into your eyes, flush them in the eyewash; hold your eyes open or have someone assist you. If you spill concentrate on skin or clothing, flush the area immediately with water. Have your lab partner notify your instructor about the spill. Silver solution will form dark spots on skin if spilled. The spots will not appear for about 24 hours, as the ions are slowly reduced to the metal. They are not hazardous, and will fade in a few days.Waste Disposal
All of the solutions prepared in this experiment should be discarded in the waste container on the bench. You may wish to have a beaker in your work area to collect waste while you are doing the experiment. Make sure it is labeled. Use a squeeze bottle of deionized water to rinse the solutions into the beaker; use a minimum amount of water to avoid creating large volumes of waste solution. The plates and test tubes can then be washed in the normal manner.Lab Procedure
Please print the worksheet for this lab. You will need this sheet to record your data.Part A: Fe3+ + SCN- 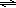 FeSCN2+ Equilibrium
FeSCN2+ Equilibrium
-
1In five wells on a ceramic spot plate, place 2 drops of 0.05 M NaSCN (sodium thiocyanate), 2 drops of 0.01 M Fe(NO3)3 solution, and 3 drops of deionized water. Make sure you have taken the correct concentrations of each solution. Mix each with a stirring rod; all of the solutions should appear red.
-
2Add 2 drops of deionized water to well 1. This well will serve as your color comparison for the following experiments.
-
3Add 2 drops of 0.10 M Fe(NO3)3 to Well 2. Record your observations in Table A.
-
4Add 2 drops of 0.05 M NaSCN to Well 3. Record your observations in Table A.
-
5Add 1 drop of 1.0 M AgNO3 to Well 4. Record your observations in Table A.
-
6Add 1 drop of 1.0 M NaNO3 to Well 5. Record your observations in Table A.
-
7After answering the questions below, rinse the contents of wells 1 - 5 into your waste beaker with a minimum amount of deionized water.
Part B: CoCl42- + 6 H2O 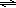 Co(H2O)62+ + 4 Cl- Equilibrium
Co(H2O)62+ + 4 Cl- Equilibrium
-
1In each of two wells of a ceramic spot plate, place 1 drop of 0.10 M Co(NO3)2.
-
2Under the HOOD, add 12 M HCl (CAUTION) dropwise to Well 1, with gentle mixing (use a stirring rod) until a distinct color change occurs. Record your observations in Table B as "Well 1A". Now add deionized water dropwise to the same well until another color change occurs. Record this observation in Table B as "Well 1B".
-
3Under the HOOD, add 12 M HCl (CAUTION) dropwise to Well 2, with gentle mixing (use a stirring rod) until a distinct color change occurs, as you did before. Record your observations in Table B as "Well 2A". The result will be similar to that in Step 2, but note it anyway. Next, add 3 drops of 1.0 M AgNO3 dropwise, with mixing, to the same well. Record your observations in Table B as "Well 2B".
-
4Place about 15 mL of the pink cobalt solution in a 30 mL beaker. Under the hood, add 12 M HCl until the solution is purple in color. Put half of the solution in a second 30 mL beaker. Warm one of the beakers on a hot plate, and cool the second beaker in an ice bath. Record any color changes that you observe in Table B.
-
5After answering the questions below, rinse the contents of the well plate and the two beakers into your waste beaker with a minimum amount of deionized water. Empty your waste beaker into the waste container provided on the side shelf, rinsing with a minimum amount of deionized water. Clean and dry all your equipment and return it to the set-up area where you found it.
-
6Before leaving, go to a computer in the laboratory and enter your results in the In-Lab assignment. If all results are scored as correct, log out. If not all results are correct, try to find the error or consult with your lab instructor. When all results are correct, note them and log out of WebAssign. The In-Lab assignment must be completed by the end of the lab period. If additional time is required, please consult with your lab instructor.